2026-04-13 中国科学院(CAS)
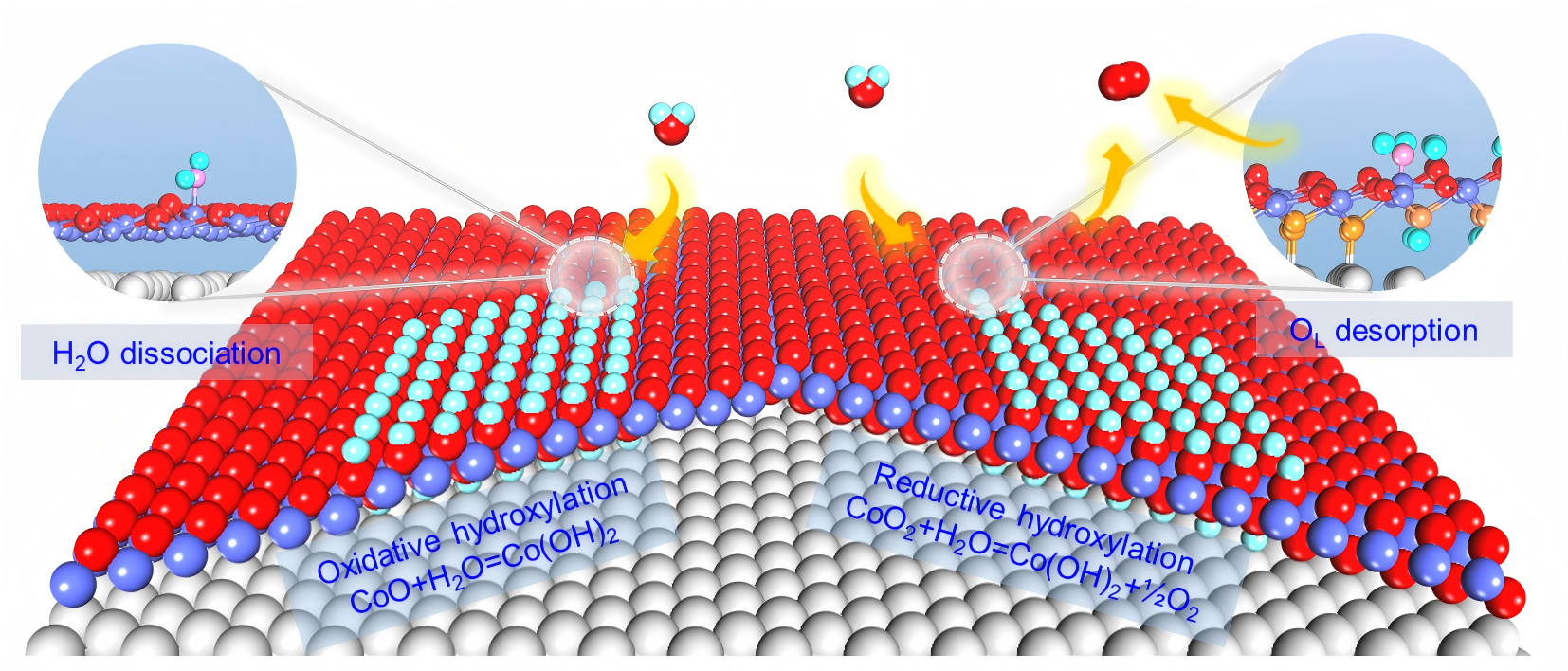
Schematic of reductive and oxidative hydroxylation of CoOx nanostructures in H2O atmosphere. (Image by SUN Xiaoyuan)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202604/t20260414_1156148.shtml
- https://academic.oup.com/nsr/article/13/6/nwag085/8469403?login=false
水蒸気中における CoO xナノ構造 の還元および酸化水酸化の動的観察 Dynamic observation of reductive and oxidative hydroxylation of CoOx nanostructures in water vapor
Xiaoyuan Sun ,Dongqing Wang ,Rankun Zhang ,Xiaoyu Liang ,Le Lin ,Rongtan Li ,Rentao Mu ,Qiang Fu
National Science Review Published:09 February 2026
DOI:https://doi.org/10.1093/nsr/nwag085
ABSTRACT
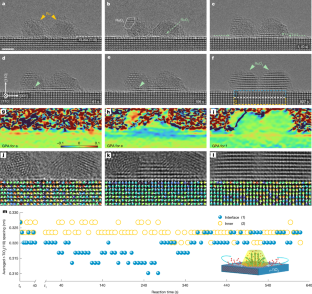
Water plays important roles in many energy chemistry and catalysis processes and yet atomic-scale understanding of water–solid interactions in water-involved interfacial processes still remains underexplored. Here, combining high-pressure scanning tunneling microscopy and theoretical calculation we have visualized and elucidated both oxidation of CoO bilayers and reduction of CoO2 trilayers in water atmospheres. CoO bilayers are readily hydroxylated to Co(OH)2 with slight Co oxidation at 10−8 mbar H2O. At the CoO2−x surface, containing both CoO and CoO2 domains, hydroxylation of CoO produces a metastable Co(OH)2-CoO2−x interface, where H2O assists oxygen desorption from interfacial CoO2 and further hydroxylation of newly formed CoO. The dynamic Co(OH)2-CoO2−x reaction front drives unusual reductive hydroxylation of CoO2−x into Co(OH)2 under mbar-level H2O. Both CoO2 reduction through H2O-assisted oxygen desorption and CoO oxidation via H2O dissociative adsorption reveal a dynamic redox mechanism for water–oxide interactions.