2026-04-17 分子科学研究所
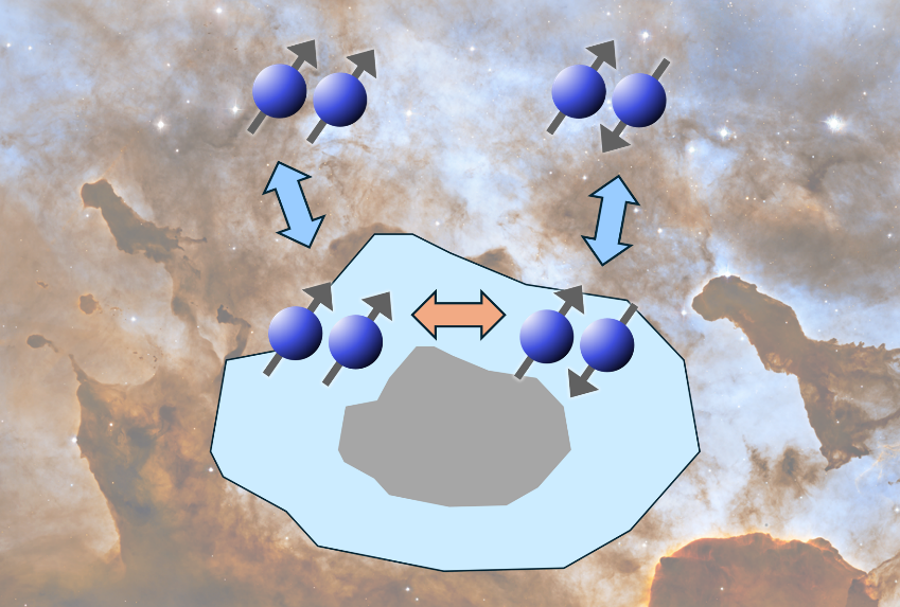
星間空間(分子雲)のダスト表面で起こる水素分子(H2)のオルソ・パラ核スピン転換(背景画像は©NASA)
<関連情報>
- https://www.ims.ac.jp/news/2026/04/0417.html
- https://iopscience.iop.org/article/10.3847/1538-4357/ae43e3
不均一な結晶粒表面におけるH₂オルト-パラスピン変換。II. 吸着オルト-H₂とパラ-H₂の回転エネルギー差の影響と重水素分画化学への示唆 H2 Ortho–Para Spin Conversion on Inhomogeneous Grain Surfaces. II. Impact of the Rotational Energy Difference between Adsorbed Ortho-H2 and Para-H2 and Implication for Deuterium Fractionation Chemistry
Kenji Furuya, Toshiki Sugimoto, Kazunari Iwasaki, Masashi Tsuge, and Naoki Watanabe
The Astrophysical Journal Published: 2026 April 6
DOI:10.3847/1538-4357/ae43e3
Abstract
We investigate how the H2 ortho-to-para ratio (OPR) and deuterium fractionation in star-forming regions are affected by nuclear spin conversion (NSC) on dust grains. Particular focus is placed on the rotational energy difference between ortho-H2 (o-H2) and para-H2 (p-H2) on grain surfaces. While the ground state of o-H2 has a higher rotational energy than that of p-H2 by 170.5 K in the gas phase, this energy difference is expected to become smaller on solid surfaces, where interactions between the surface and adsorbed H2 molecules affect their rotational motion. A previous study by K. Furuya et al. developed a rigorous formulation of the rate for the temporal variation of the H2 OPR via the NSC on grains, assuming that adsorbed o-H2 has higher rotational energy than adsorbed p-H2 by 170.5 K, as in the gas phase. In this work, we relax the assumption and reevaluate the rate, varying the rotational energy difference between their ground states. The reevaluated rate is incorporated into a gas-ice astrochemical model to study the evolution of the H2 OPR and the deuterium fractionation in prestellar cores and the outer, cold regions of protostellar envelopes. The inclusion of the NSC on grains reduces the timescale of the H2 OPR evolution and thus the deuterium fractionation, at densities of ≳104 cm−3 and temperatures of ≲14–16 K (depending on the rotational energy difference), when the ionization rate of H2 is 10−17 s−1.