2026-03-26 東京科学大学
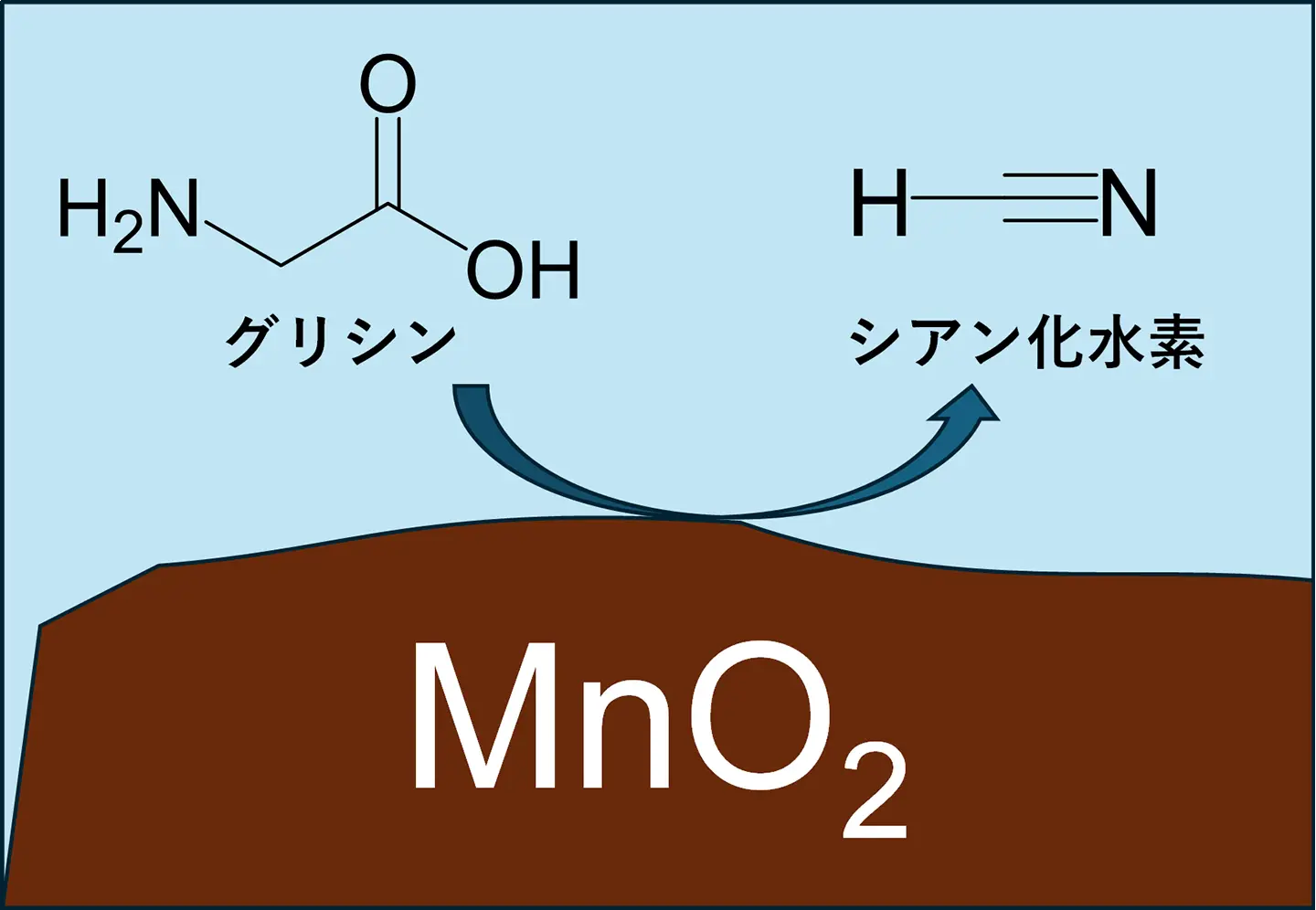
図1. 二酸化マンガンが促進するアミノ酸からシアン化水素の合成
<関連情報>
化学進化のための、前生物的に豊富に存在するアミノ酸からのシアン化水素の鉱物促進型水溶液合成 Mineral-facilitated aqueous synthesis of hydrogen cyanide from prebiotically abundant amino acids for chemical evolution
Zening Yang, Yamei Li, Norio Kitadai, +7 , and Ryuhei Nakamura
Proceedings of the National Academy of Sciences Published:March 23, 2026
DOI:https://doi.org/10.1073/pnas.2515805123
Significance
Origin of life strongly relies on reactive carbon and nitrogen precursors, among which hydrogen cyanide (HCN) is one of the most versatile molecules that can be used for synthesizing almost all essential biomolecules. However, recent geochemical studies have raised concern about the availability of HCN, as the highly reducing methane-rich atmosphere, which is required for HCN synthesis, has been considered to be uncertain. Different from methane, amino acids can be synthesized via diverse chemical pathways and supplied by extraterrestrial delivery; thus, amino acids are likely prebiotically abundant. Here, we report aqueous phase synthesis of HCN from amino acids promoted by minerals, which can address the problem of prebiotic supply of HCN on early Earth toward chemical evolution.
Abstract
Recent advances in prebiotic chemistry suggest that hydrogen cyanide (HCN) serves as a fundamental precursor for nearly all essential biomolecules and protometabolic processes for life’s emergence. Although prevailing models of endogenous cyanide production rely on atmospheric methane, the availability of methane remains uncertain. Unlike methane, amino acids were likely prebiotically abundant, forming through multiple synthetic pathways under a methane-free, nonreducing atmosphere. Here, we demonstrate that HCN can form from proteinogenic amino acids in anaerobic aqueous solutions facilitated by geochemically available minerals, offering a route to overcome HCN scarcity under a nonreducing atmosphere. On manganese dioxide, the glycine-to-cyanide conversion proceeded across a broad range of pHs (2.0 to 12.6) and substrate concentrations (1 μM to 100 mM), achieving a maximum selectivity of 57%. The reaction involves α-proton abstraction in amino acids, which is distinct from conventional chemical decarboxylation processes of amino acids, highlighting the unique role of MnO2 in activating the α-C-H bond to form HCN. HCN generation was observed for nearly all proteinogenic amino acids and short peptides. Because amino acids can be synthesized from HCN-independent pathways from abundant carbon sources such as CO2 and CO, amino acid-derived HCN alleviates the requirement on atmospheric methane and can sustain continued chemical evolution in ambient aqueous environments.