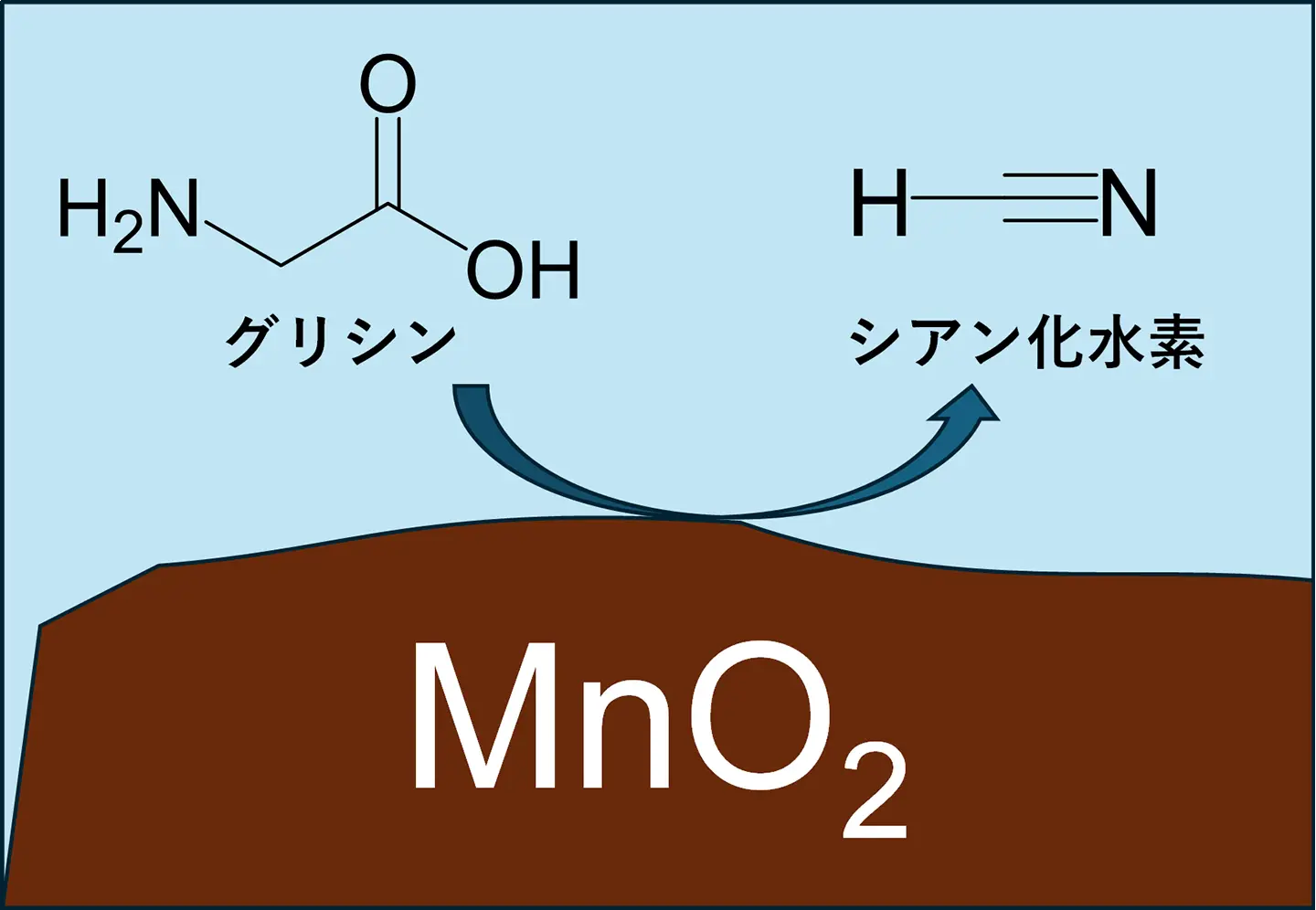
2026-03-26 京都大学,科学技術振興機構
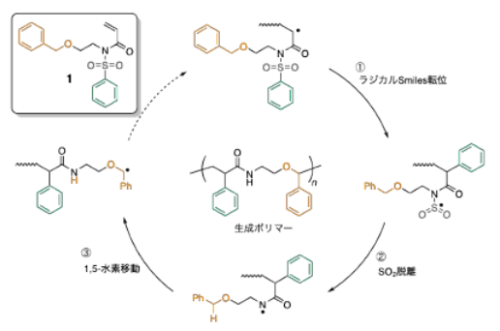
図1 カスケード型ラジカル異性化重合による主鎖にアミド結合を周期的に導入した高分子の合成
<関連情報>
- https://www.jst.go.jp/pr/announce/20260326/index.html
- https://www.jst.go.jp/pr/announce/20260326/pdf/20260326.pdf
- https://pubs.acs.org/doi/10.1021/jacs.5c21559
カスケードラジカル異性化重合によるポリマー骨格の設計 Cascade Radical Isomerization Polymerization to Engineer Polymer Backbones
Keita Kuroda,Makoto Ouchi
Journal of the American chemistry Society Published: March 23, 2026
DOI:https://doi.org/10.1021/jacs.5c21559
Abstract
We designed disubstituted acrylamide monomers bearing an arylsulfonyl substituent and an alkyl substituent containing a radical-stabilizing group (i.e., benzene) to produce polymers whose backbones incorporate amide linkages through radical migration (isomerization) during polymerization. The monomer structure was engineered so that, after radical addition to the vinyl group, a 1,4-Smiles rearrangement to the arylsulfonyl group, SO2 extrusion, and intramolecular 1,5-hydrogen atom transfer to the benzyl group would proceed sequentially, generating a nucleophilic benzyl radical capable of propagating with the electron-deficient vinyl group of the monomer. By varying the polymerization conditions and conducting detailed NMR structural analyses of the resulting polymers, we achieved up to 92% incorporation of the repeat unit formed through the cascade isomerization among all repeating units. Moreover, structural modification of the monomer framework with diverse substituents allowed tuning of the glass-transition temperature (Tg) with the cascade-unit content and imparted a degradable character under acidic conditions through incorporation of an ether linkage in the polymer backbone.