2026-04-28 合肥物質科学研究院(HFIPS)
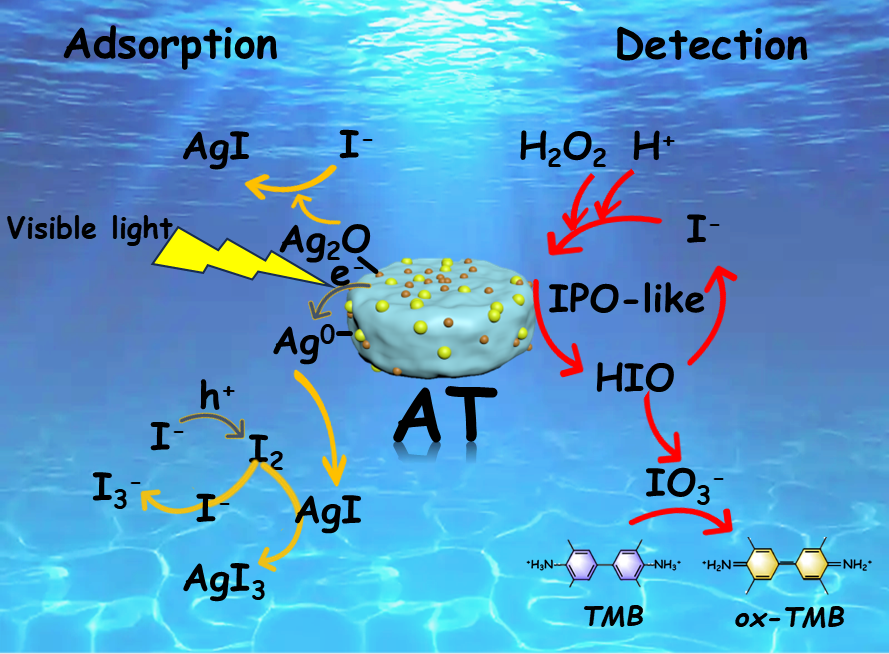
Adsorption and detection mechanism of radioactive iodide ions by AT nanocomposites. (Image by CHEN Kaiwen)
<関連情報>
- https://english.hf.cas.cn/nr/rn/202604/t20260428_1158201.html
- https://www.sciencedirect.com/science/article/abs/pii/S1383586626008968
MIL-125由来の銀修飾酸素空孔リッチTiO2 -xによる微量放射性ヨウ化物の効率的な除去と肉眼検出Efficient elimination and naked-eye detection of trace radioactive iodide by silver-decorated oxygen vacancy-rich TiO2-x derived from MIL-125
Kai-Wei Chen, Chao Wu, Ming-Jun Zhang, Mohamed Mitwalli, Yue-Yue Liu, Hai-Xia Wang, Tao-Sheng Li, Yi Yang
Separation and Purification Technology Available online: 16 March 2026
DOI:https://doi.org/10.1016/j.seppur.2026.137630
Highlights
- Ag2O-Ag@TiO2-x was constructed by pyrolysis-reduction using MIL-125 as a precursor.
- The adsorption capacity of AT for iodide is up to 143.9 mg g−1 under visible-light.
- AT achieved enhanced removal of iodide through photocatalysis-oxidation process.
- I2 generated by oxidation was captured to form AgI, and further formed AgI3 or I3−.
- AT detected iodide with high sensitivity based on its IPO activity (LOD: 1.33 μ M).
Abstract
The development of efficient radioactive iodine management technologies is crucial for ensuring the sustainable utilization of nuclear energy. Herein, silver nanoparticle-modified oxygen-rich vacancy TiO2-x (Ag2O-Ag@TiO2-x, AT) nanocomposites were successfully synthesized via a controlled pyrolysis-reduction strategy using a Ti-based MOF (MIL-125) as the precursor. The synergistic effect of oxygen vacancies and Ag/TiO2-x Schottky junctions endowed the material with exceptional iodide adsorption performance through a photocatalytic oxidation-adsorption mechanism, achieving a maximum adsorption capacity of 143.9 mg g−1. Notably, the AT nanocomposites maintained high adsorption efficiency (removal rate > 95%) even for trace-level iodide ions (down to 300 μg L−1). Furthermore, the material exhibited superior iodoperoxidase-like activity, enabling highly sensitive colorimetric detection of iodide with a detection limit of 1.33 μM. This sensing mechanism relies on H2O2-mediated oxidation of I− to reactive iodine species (IO3−), which subsequently convert 3,3′,5,5′-tetramethylbenzidine (TMB) into oxidized TMB (ox-TMB). Combined experimental and theoretical analyses elucidated the underlying mechanisms governing iodide adsorption and sensing. This work not only provided fundamental insights into iodine-material interactions but also offered a practical solution for environmental monitoring and nuclear waste management, effectively addressing critical challenges in radioactive iodine remediation.