2026-03-12 中国科学院(CAS)
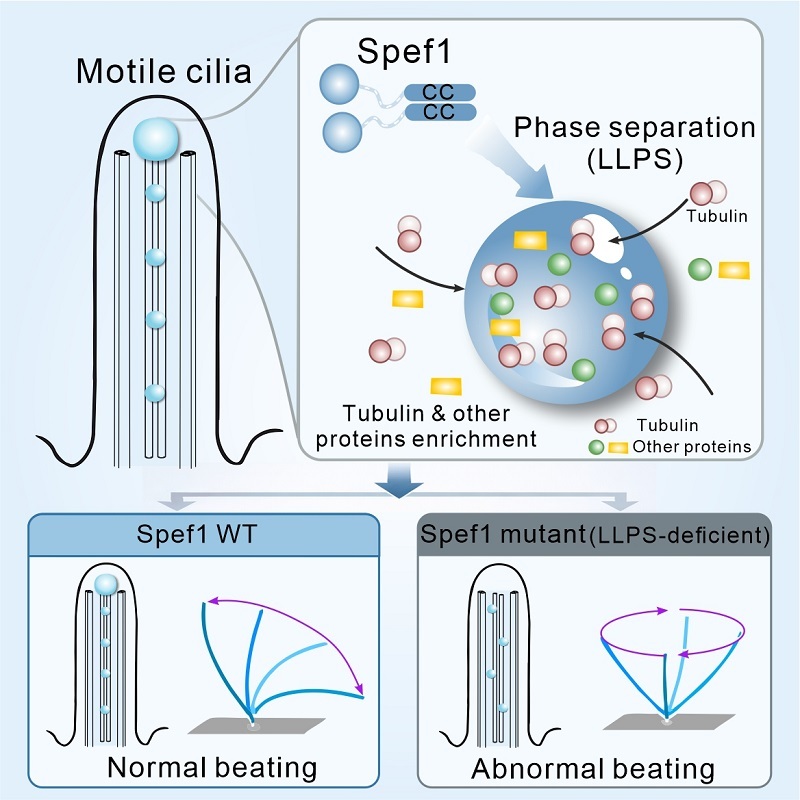
Schematic model illustrating how Spef1 undergoes phase separation to modulate ciliary beating. (Image by FENG Wei’s group)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202603/t20260312_1152618.shtml
- https://www.cell.com/structure/abstract/S0969-2126(26)00049-3
中心対微小管の組織化と機能のためのコイルドコイルを介したSpef1の相分離 Coiled-coil-mediated phase separation of Spef1 for central-pair microtubule organization and function
Jinqi Ren ∙ Juyuan Liu ∙ Kewei Gu ∙ … ∙ Benhua Qiu ∙ Xueliang Zhu ∙ Wei Feng
Structure Published:March 10, 2026
DOI:https://doi.org/10.1016/j.str.2026.02.007
Highlights
- Spef1 undergoes liquid-liquid phase separation both in vitro and in cells
- Spef1 droplets enrich tubulins and promote microtubule assembly
- A C-terminal coiled-coil domain mediates Spef1 dimerization and phase separation
- Loss of Spef1 liquid-liquid phase separation leads to defective ciliary beating
Summary
Central-pair microtubules (CP-MTs) are non-centrosomal MTs essential for planar beat pattern of cilia. The CP-MT formation requires the MT-associated protein Spef1, but the underlying molecular mechanism remains unclear. Here, we show that Spef1 undergoes liquid-liquid phase separation (LLPS) to facilitate non-centrosomal MT assembly by enriching tubulins. The LLPS of Spef1 is mediated by its C-terminal coiled-coil (CC) domain. Crystallography reveals that the Spef1-CC domain forms a parallel CC dimer with a unique charge distribution pattern on the surface. The dimerization capacity and charge distribution of Spef1-CC are both critical for controlling invitro LLPS. Disruption of the dimerization capacity abolishes ciliary functions of Spef1. In contrast, a charge-changing mutant with attenuated LLPS still supports the CP-MT formation but results in cilia with abnormal beat pattern. Thus, the CC-mediated LLPS of Spef1 provides a mechanistic explanation for its prominent role in controlling CP-MT organization and function in the axoneme.



