2025-04-17 ワシントン大学セントルイス校
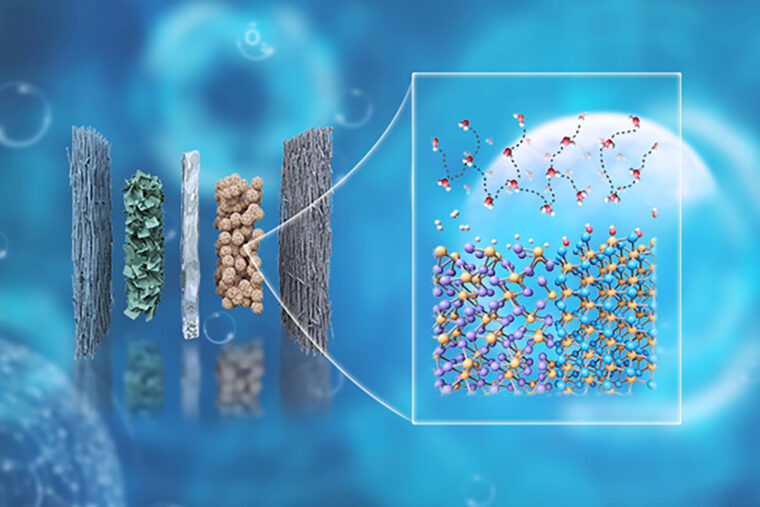
A team of researchers led by Gang Wu created a new energy-efficient catalyst using two phosphides to split hydrogen from water. The image on the left shows the dry cathode anion-exchange membrane water electrolyzer, and the image on the right shows the connected dynamic hydrogen bond network. (Image: Gang Wu)
<関連情報>
- https://source.washu.edu/2026/04/washu-researchers-use-efficient-method-to-split-hydrogen-from-water-for-energy/
- https://engineering.washu.edu/news/2026/WashU-researchers-use-efficient-method-to-split-hydrogen-from-water-for-energy.html
- https://pubs.acs.org/doi/10.1021/jacs.6c02768
アルカリ水電解用リン化物ヘテロ構造/電解質界面における水素結合ネットワーク制御による乾式陰極の設計 Designing a Dry Cathode via Hydrogen-Bond Network Regulation at Phosphide Heterostructure/Electrolyte Interfaces for Alkaline Water Electrolysis
Jiashun Liang,Yu Li,Chun-Wai Chang,Mingxuan Qiao,Zhenxing Feng,Chaochao Dun,Wan-Lu Li,and Gang Wu
Journal of the American Chemical Society
DOI:https://doi.org/10.1021/jacs.6c02768
Abstract
Developing efficient platinum group metal (PGM)-free catalysts for the hydrogen evolution reaction (HER) in alkaline electrolytes is crucial for anion-exchange membrane water electrolyzers (AEMWEs) to produce low-cost hydrogen. Despite the promising activity often observed in aqueous electrolytes on HER catalysts, catalytic performance in the actual AEMWE cathode environment is primarily limited by insufficient water and proton supply under desirable dry-cathode operating conditions. Herein, we develop a Re2P/MoP heterostructure catalyst that overcomes these mass-transport and interfacial limitations, enabling a high-performance PGM-free cathode in AEMWEs. Relative to the individual Re2P and MoP counterparts, the Re2P/MoP heterostructure catalyst exhibits significantly lower HER overpotentials. Electrochemical measurements and theoretical calculations suggest that coupling between Re2P and MoP can regulate the electronic structure of each component, thereby optimizing hydrogen adsorption and water dissociation kinetics and enhancing intrinsic HER activity. Moreover, the Re2P/MoP heterostructure can populate interfacial water molecules and form a connected/dynamic hydrogen-bond network near the catalyst/electrolyte interface, thereby facilitating water replenishment and proton/hydroxide transfer, which are essential for high-current-density operations even at low water content. The assembled Re2P/MoP||NiFe foam PGM-free membrane electrode assembly (MEA) delivers an industrial-level current density of 1.0 and 3.0 A cm–2 at 1.73 and 1.95 V, respectively, comparable to the PtRu/C||IrOx PGM benchmark. The PGM-free MEA can maintain stable operation at 2.0 A cm–2 for over 1000 h, making it one of the most promising MEAs for AEMWEs. This work emphasizes that catalyst/electrolyte interface engineering is critical for achieving high-performance electrochemical systems.



