2026-03-31 東京科学大学
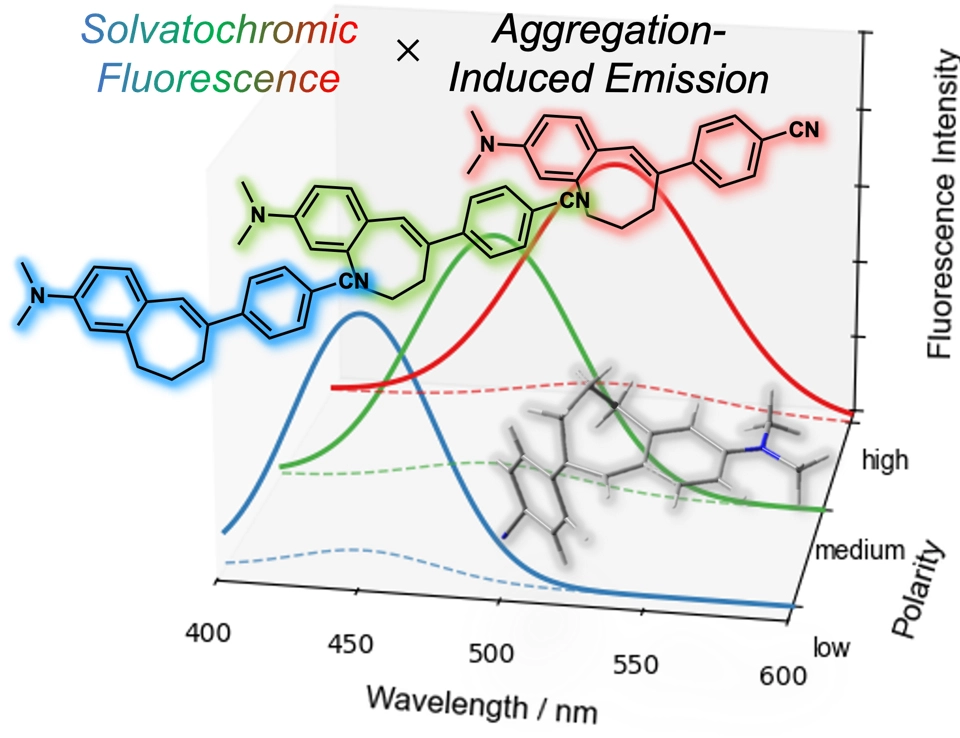
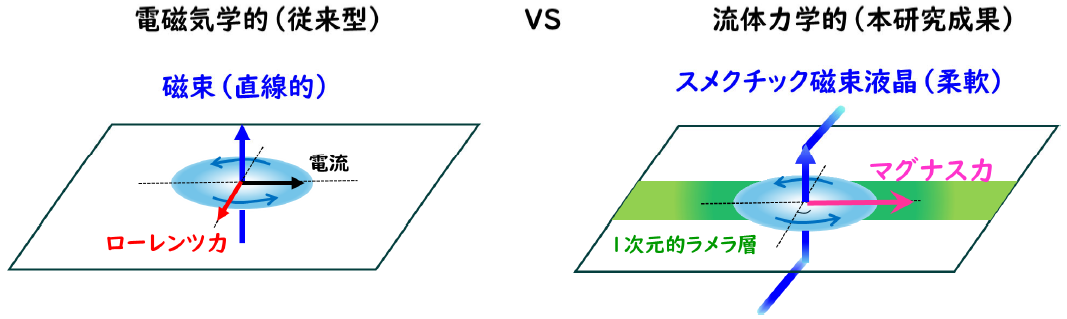
図. 本研究の概要
<関連情報>
- https://www.isct.ac.jp/ja/news/30lfkdw8rh1h#top
- https://onlinelibrary.wiley.com/doi/10.1002/agt2.70295
Push-Pull型橋かけスチルベンを基盤とした小型ソルバトクロミック凝集誘起発光色素の設計と励起状態失活ダイナミクス Push–Pull Bridged Stilbenes as Small Solvatochromic Aggregation-Induced Emission Luminogen: Design and Excited-state Deactivation Dynamics
Takuya Tanaka, Hirosato Koyanagi, Takumi Ehara, Tomohiro Ryu, Kiyoshi Miyata, Satoshi Suzuki, Kazunobu Igawa, Ken Onda, Gen-ichi Konishi
Aggregate Published: 14 February 2026
DOI:https://doi.org/10.1002/agt2.70295
ABSTRACT
Aggregation-induced emission luminogens (AIEgens) are typically large π-conjugated molecules, but their low affinity and noninvasiveness toward analytes limit practical applications. To address this, smaller, more planar AIEgens are needed. Stilbene, though structurally suitable, lacks visible luminescence. Here, we report a minimally modified stilbene-based AIEgen—4-dipropylamino-4’-cyano-bridged stilbene (DpCBS[7])—that exhibits fluorescence solvatochromism and efficient AIE across a broad polarity range in the visible region. DpCBS[7] exhibits low quantum yields (Φfl = 0.01–0.04) in solvents from nonpolar n-hexane to polar dimethyl sulfoxide, with large Stokes shifts, viscosity-sensitive luminescence, and highly efficient solid-state luminescence (Φfl = 0.70). To elucidate its dual solvatochromic and AIE behavior, femtosecond transient absorption spectroscopy was conducted. In solution, DpCBS[7] displays transient absorption with lifetimes of 21 ps (toluene) and 56 ps (acetonitrile) at 293 K, indicating ultrafast nonradiative decay leading to low Φfl. Arrhenius analysis over the temperature range of 263–313 K revealed activation energies (ΔEa) of 9.90 kJ/mol in toluene and 12.8 kJ/mol in acetonitrile for the S1 → S0 decay of DpCBS[7]. The ΔEa values show no clear systematic dependence on solvent polarity. In contrast, pre-exponential factor A remains consistently high regardless of solvent polarity, indicating that the striking photophysical response is governed primarily by the pre-exponential factor rather than by modulation of the activation energy. These findings highlight the fundamental importance of tailoring the distribution function through structural modification as a robust strategy to control AIE characteristics.