2026-04-02 北海道大学
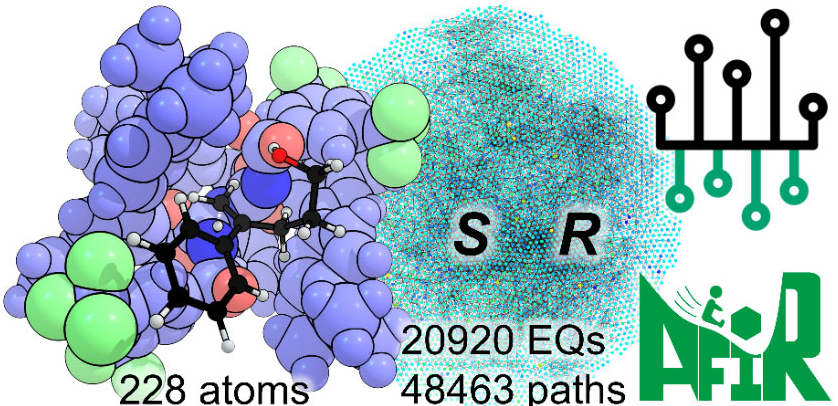
巨大反応経路ネットワークを用いた不斉触媒反応予測の概念図。
<関連情報>
- https://www.hokudai.ac.jp/news/2026/04/post-2239.html
- https://www.hokudai.ac.jp/news/pdf/260402_pr2.pdf
- https://pubs.acs.org/doi/10.1021/acscentsci.6c00079
巨大反応経路ネットワークにおける速度論シミュレーションによるエナンチオ選択性の予測 Predicting Enantioselectivity via Kinetic Simulations on Gigantic Reaction Path Networks
Yu Harabuchi,Ruben Staub,Min Gao,Nobuya Tsuji,Benjamin List,Alexandre Varnek,and Satoshi Maeda
ACS Sentral Science Published: March 30, 2026
DOI:https://doi.org/10.1021/acscentsci.6c00079
Abstract
Asymmetric catalysts exert control over the reactivity and enantioselectivity of chemical reactions, often through their large size and highly flexible geometries. Most computational approaches rely on a predefined selectivity-determining step and assume that selectivity follows the Boltzmann distribution of transition-state conformers. Going beyond this conventional framework, we constructed a reaction path network that captures the kinetically accessible regions of the potential energy surface. A delta-learning neural network potential (ΔNNP) was constructed and applied to an asymmetric organocatalyst of the imidodiphosphorimidates family, comprising over 200 atoms. The ΔNNP achieved DFT-level accuracy with GFN2-xTB as the baseline. Using the ΔNNP-based single component-artificial force induced reaction (denoted by NNP/AFIR) method, we constructed a reaction path network containing 48,463 paths. Kinetic simulations based on this network predicted the reaction mechanism and enantioselectivity. Within the network, numerous paths, including asynchronous concerted and stepwise mechanisms, were found to be energetically competitive and thus to contribute to the overall kinetics. Traffic volume analysis further revealed that intermediates with negligible yields can still play important kinetic roles. Overall, the NNP/AFIR approach provides a powerful framework not only for deepening mechanistic understanding but also for accelerating the rational design of asymmetric catalytic systems with high reactivity and enantioselectivity.



