水の電気分解に光が新たな電極触媒として作用することを発見し、クリーンエネルギーである水素をより安価に入手できるようになる可能性がある。 The team’s discovery that light can trigger a brand new electro-catalytic mechanism of water electrolysis could improve affordability of hydrogen as source of clean energy
2022-10-27 シンガポール国立大学(NUS)
約3年前に偶然にも研究室の天井の照明が停電し、暗闇の中で続けていたオキシ水酸化ニッケル系材料の水の電気分解実験の性能が大幅に低下していたのです。
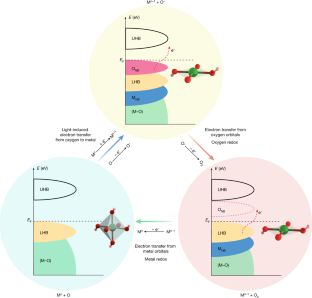
Xue准教授は、「私たちは、電気触媒反応の酸化還元中心が、光によって金属と酸素の間で切り替わることを発見しました。これは水の電気分解の効率を大きく向上させるものです。」と述べました。
この発見について、2022年10月26日に科学雑誌「Nature」に掲載された研究論文に詳述しています。
<関連情報>
- https://news.nus.edu.sg/revolutionary-technique-to-generate-hydrogen-more-efficiently-from-water/
- https://www.nature.com/articles/s41586-022-05296-7
酸素発生における可逆的なNiO6幾何学変換の重要な役割 Pivotal role of reversible NiO6 geometric conversion in oxygen evolution
Xiaopeng Wang,Shibo Xi,Pengru Huang,Yonghua Du,Haoyin Zhong,Qing Wang,Armando Borgna,Yong-Wei Zhang,Zhenbo Wang,Hao Wang,Zhi Gen Yu,Wee Siang Vincent Lee & Junmin Xue
Nature Published:26 October 2022
DOI:https://doi.org/10.1038/s41586-022-05296-7
Abstract
Realizing an efficient electron transfer process in the oxygen evolution reaction by modifying the electronic states around the Fermi level is crucial in developing high-performing and robust electrocatalysts. Typically, electron transfer proceeds solely through either a metal redox chemistry (an adsorbate evolution mechanism (AEM), with metal bands around the Fermi level) or an oxygen redox chemistry (a lattice oxygen oxidation mechanism (LOM), with oxygen bands around the Fermi level), without the concurrent occurrence of both metal and oxygen redox chemistries in the same electron transfer pathway. Here we report an electron transfer mechanism that involves a switchable metal and oxygen redox chemistry in nickel-oxyhydroxide-based materials with light as the trigger. In contrast to the traditional AEM and LOM, the proposed light-triggered coupled oxygen evolution mechanism requires the unit cell to undergo reversible geometric conversion between octahedron (NiO6) and square planar (NiO4) to achieve electronic states (around the Fermi level) with alternative metal and oxygen characters throughout the oxygen evolution process. Utilizing this electron transfer pathway can bypass the potential limiting steps, that is, oxygen–oxygen bonding in AEM and deprotonation in LOM. As a result, the electrocatalysts that operate through this route show superior activity compared with previously reported electrocatalysts. Thus, it is expected that the proposed light-triggered coupled oxygen evolution mechanism adds a layer of understanding to the oxygen evolution research scene.

 ビーチクラフトA36、春秋航空公司エアバスA320の重大インシデント[他の航空機との衝突又は接触のおそれ](佐賀空港西南西上空、令和元年12月21日発生)](https://tiisys.com/wp-content/uploads/2022/10/aki-150x136.png)
