2026-03-03 大阪大学,科学技術振興機構
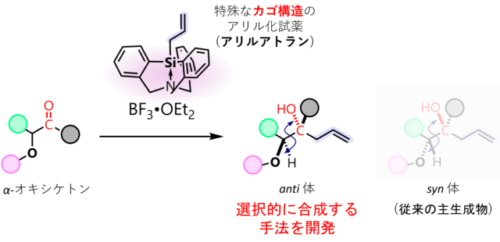
図 1. カゴ構造のアリル化試薬を用いた従来にない空間配置をもつアリル化の実現
<関連情報>
- https://www.jst.go.jp/pr/announce/20260303/index.html
- https://www.jst.go.jp/pr/announce/20260303/pdf/20260303.pdf
- https://www.nature.com/articles/s41467-026-69732-2
14族アリルトランを用いたα-オキシケトンのアリル化における非キレート化制御 Non-chelation control in allylations of α-oxy ketones using group-14 allylatranes
Yuya Tsutsui,Kokoro Shiga,Akihito Konishi & Makoto Yasuda
Nature Communications Published:03 March 2026
DOI:https://doi.org/10.1038/s41467-026-69732-2
Abstract
Stereoselective nucleophilic additions to α-substituted carbonyl compounds are a crucial area of contemporary research in organic chemistry. Of the various advancements in π-facial selectivity in addition reactions of carbonyl compounds, the (polar) Felkin-Anh model and the chelation model are well recognized for accurately explaining the selectivity of the allylic products. For reactions that involve α-oxy carbonyl groups – known for their broad applications in natural-product synthesis and as effective building blocks in organic synthesis – the stereoselective reaction typically follows the chelation model, favoring syn-selective addition. In contrast to the well-established syn-selective additions of α-oxy carbonyls, anti-selective additions through a non-chelation pathway remain largely unexplored. In this study, we present the anti-selective allylation of α-oxy ketones using allylatranes that feature a highly coordinated group-14-element center. These atranes demonstrate high nucleophilicity and low chelating ability due to their transannular interactions and rigid framework, facilitating anti-selective allylations. A combined experimental and theoretical approach has been used to highlight the unique electronic properties of these atranes. This method is applicable to a wide variety of substrates, producing anti-1,2-diols with a homoallylic moiety in high yield and excellent diastereoselectivity compared to traditional methods.