2026-02-20 ゲーテ大学
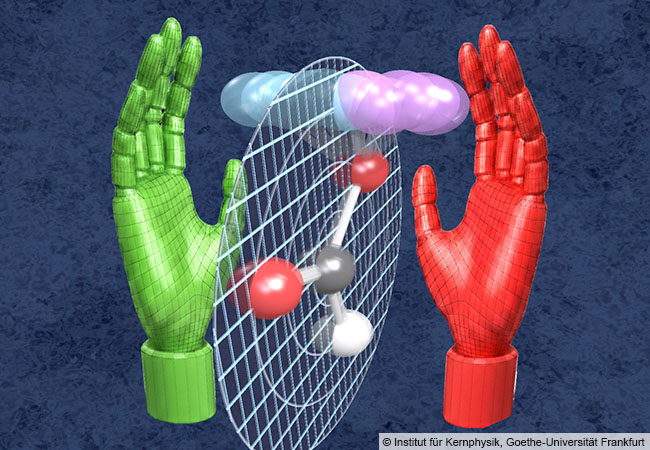
Quantum mechanical zero-point vibration—the “trembling” of the atoms—makes formic acid a chiral molecule whose two forms, like the right and left hand, cannot be superimposed. © Institute for Nuclear Physics, Goethe University Frankfurt
<関連情報>
- https://aktuelles.uni-frankfurt.de/english/the-quantum-trembling-why-there-are-no-truly-flat-molecules/
- https://journals.aps.org/prl/abstract/10.1103/bvqj-pm3n
ギ酸の平面基底状態における瞬間的な単一分子キラリティーの探査 Probing Instantaneous Single-Molecule Chirality in the Planar Ground State of Formic Acid
D. Tsitsonis, M. Kircher, N. M. Novikovskiy, F. Trinter, J. B. Williams, K. Fehre, L. Kaiser, S. Eckart, O. Kreuz et al.
Physical Review Letters Published: 30 January, 2026
DOI: https://doi.org/10.1103/bvqj-pm3n
Abstract
We experimentally demonstrate that individual molecules of formic acid are chiral even when they are in the vibronic ground state, which has a planar equilibrium structure. We ionize the C 1s shell of the molecule and record the photoelectron in coincidence with positively charged fragments. This provides two consecutive measurements of the structure of one molecule, the first by photoelectron diffraction imaging and the second by Coulomb explosion imaging. We find that both measurements show the same handedness of the specific molecule. The phenomenon of being achiral on average but chiral at the level of individual molecules is general to prochiral molecules and is a consequence of the three-dimensional zero-point delocalization of the nuclei in the vibrational ground state.