2026-05-12 ロッキーズ国立研究所(NLR)
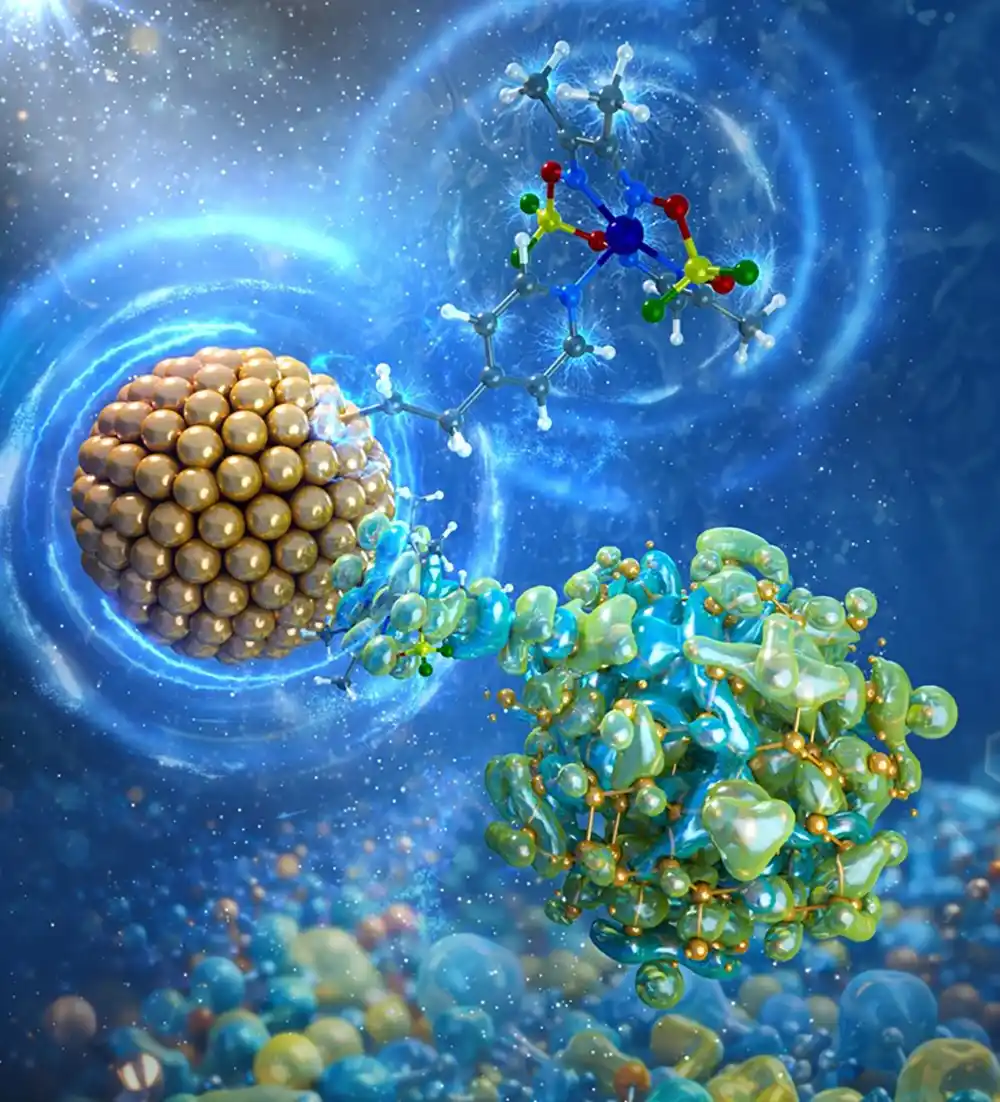
A molecular catalyst (multicolored ball-and-stick model) and silicon nanocrystal (sphere made up of gold balls) form hybrid states (green and blue mixture linked to the silicon nanocrystal). These enable high-energy charges to persist for an exceptionally long time (blue radiant energy circles coming off the catalyst and silicon nanocrystal). Illustration by Joshua Bauer, Trung Le, and Nathan Neale, National Laboratory of the Rockies
<関連情報>
- https://www.nlr.gov/news/detail/program/2026/scientists-discover-way-to-leverage-high-energy-sunlight-for-fuel-production
- https://pubs.acs.org/doi/10.1021/jacs.5c19326?ref=PDF
高エネルギー混成状態により、コバロキシム-シリコンナノ結晶系において長寿命のホットエレクトロンが実現する High-Energy Hybridized States Enable Long-Lived Hot Electrons in Cobaloxime-Silicon Nanocrystal System
Trung H. Le,Melissa K. Gish,Simran S. Saund,Taylor Aubry,Nathan R. Neale
Journal of the American Chemical Society Published: February 8, 2026
DOI:https://doi.org/10.1021/jacs.5c19326
Abstract
Strong electronic coupling is achieved between the molecular catalyst cobaloxime ([Co]) and silicon nanocrystals (Si NCs) bridged by an ethylenepyridine group derived from vinylpyridine (vpy) covalently bound to the Si NC surface (Si-vpy-[Co]). The ethylenepyridine tether in Si-vpy-[Co] is key to dramatic changes to the system’s physical properties─which are not observed in the corresponding formylpyridine (fpy) system (Si-fpy-[Co])─consistent with strong electronic coupling previously observed only in dark electrochemical systems. UV–vis absorption spectroscopy reveals new [Co]-centered electronic states in Si-vpy-[Co], and transient absorption spectroscopy finds a strong absorption feature appearing within 250 fs and persisting for at least 5 ns. Astoundingly, spectroelectrochemical measurements reveal that this absorption feature is consistent with both the singly reduced [Co]− and doubly reduced [Co]2– complexes, leading to the conclusion that these long-lived charges are derived from high-energy “hot” electrons residing in [Co]-centered states. Detailed analysis using cyclic voltammetry, spectroelectrochemistry, electron paramagnetic resonance spectroscopy, and density functional theory (DFT) calculations provides insight into the unique electronic structure created in Si-vpy-[Co]. DFT reveals that the new electronic states arise from hybridization between deep Si NC band states and high-energy molecular orbitals of the ethylenepyridine tether and the [Co] catalyst and are facilitated by σ-bonding character at the ethylenepyridine linkage. This study demonstrates that strong electronic coupling achieved through precise molecular chemistry can change the paradigm of otherwise fixed energy levels in hybrid photoelectrochemical systems for artificial photosynthesis and related applications.



