2024-10-09 ウースター工科大学(WPI)
<関連情報>
- https://www.wpi.edu/news/announcements/unlocking-high-capacity-and-reversible-alkaline-iron-redox-using-silicate-sodium-hydroxide-hybrid
- https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/cssc.202400050
シリケート・水酸化ナトリウムハイブリッド電解質を用いた高容量・可逆性アルカリ鉄レドックスの開発 Unlocking High Capacity and Reversible Alkaline Iron Redox Using Silicate-Sodium Hydroxide Hybrid Electrolytes
Sathya Narayanan Jagadeesan, Dr. Fenghua Guo, Ranga Teja Pidathala, Dr. A. M. Milinda Abeykoon, Dr. Gihan Kwon, Dr. Daniel Olds, Prof. Dr. Badri Narayanan, Prof. Dr. Xiaowei Teng
ChemSusChem Published: 19 June 2024
DOI:https://doi.org/10.1002/cssc.202400050
Graphical Abstract
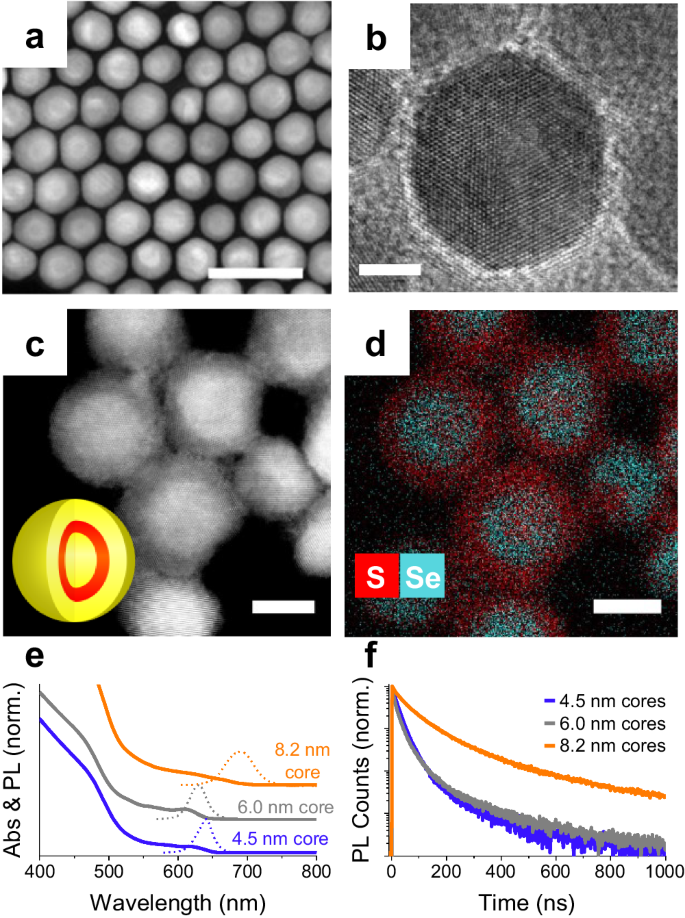
The authors report that the silicate-added hybrid electrolyte strongly interacts with iron oxide during charge to control the competing hydrogen evolution, thus improving conversion. In discharge, one electron transfer to Fe(OH)2/FeOOH conversion is achieved by mitigating electrochemically inactive Fe3O4 formation. Such innovation provides a path for designing effective aqueous electrolytes for enabling sustainable energy storage applications by repurposing iron rust.
Abstract
Alkaline iron (Fe) batteries are attractive due to the high abundance, low cost, and multiple valent states of Fe but show limited columbic efficiency and storage capacity when forming electrochemically inert Fe3O4 on discharging and parasitic H2 on charging. Herein, sodium silicate is found to promote Fe(OH)2/FeOOH against Fe(OH)2/Fe3O4 conversions. Electrochemical experiments, operando X-ray characterization, and atomistic simulations reveal that improved Fe(OH)2/FeOOH conversion originates from (i) strong interaction between sodium silicate and iron oxide and (ii) silicate-induced strengthening of hydrogen-bond networks in electrolytes that inhibits water transport. Furthermore, the silicate additive suppresses hydrogen evolution by impairing energetics of water dissociation and hydroxyl de-sorption on iron surfaces. This new silicate-assisted redox chemistry mitigates H2 and Fe3O4 formation, improving storage capacity (199 mAh g−1 in half-cells) and coulombic efficiency (94 % after 400 full-cell cycles), paving a path to realizing green battery systems built from earth-abundant materials.