2026-04-02 バース大学
<関連情報>
- https://www.bath.ac.uk/announcements/breakthrough-offers-endless-recycling-for-acrylic-plastics-without-the-environmental-cost/
- https://www.nature.com/articles/s41467-025-67997-7
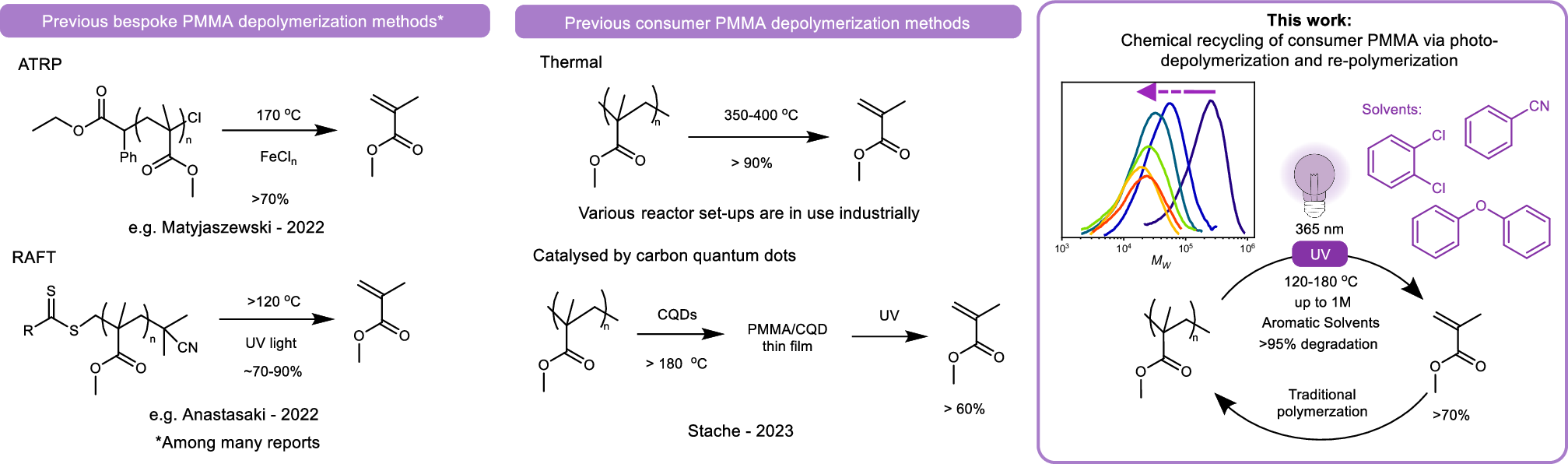
塩素系試薬を用いない、光開始溶媒媒介による消費者向けポリ(メタクリル酸メチル)の解重合 Photo-initiated solvent-mediated depolymerization of consumer poly(methyl methacrylate) without chlorinated reagents
Jonathan T. Husband,Gavin Irvine,Callum R. Morris,Andrea Folli,Matthew G. Davidson & Simon J. Freakley
Nature Communications Published:28 January 2026
DOI:https://doi.org/10.1038/s41467-025-67997-7
Abstract
The chemical recycling of commodity acrylic polymers, such as the transparent thermoplastic polymethyl methacrylate (PMMA), typically requires temperatures of 350-400°C. Herein, we report chemical recycling back to monomers for PMMA between 120-180°C, through UV illumination under oxygen-free conditions. We have achieved gram-scale degradation of consumer plastic with >95% conversion, yielding >70% monomer, which can be readily repolymerized. The process proceeds even at high concentrations (>1 M) and depends strongly on solvent choice: aromatic solvents like dichlorobenzene and diphenyl ether maximize conversion. In contrast to a concurrently published study, we report that chlorine radicals are not required for depolymerization; however, when present, they react with the unzipping chain to form chlorine-functionalized PMMA which can be upcycled through derivatization. In more sustainable non-chlorinated solvents such as benzonitrile, minimal termination by radicals enables complete unzipping. These findings demonstrate a low-temperature, scalable route for the chemical recycling of PMMA, offering alternative pathways for plastic circularity.



