2026-04-01 京都大学
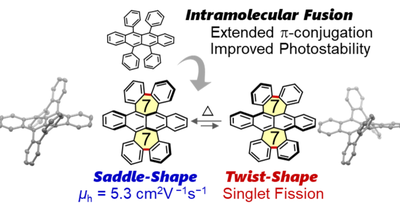
本研究の概要図:ルブレン(上)を縮環させた構造を有するFR分子(下)が 2つの配座異性体(左右)として存在することを示している(作成:久田 雅人)
<関連情報>
- https://www.t.kyoto-u.ac.jp/ja/research/topics/20260401
- https://www.t.kyoto-u.ac.jp/ja/research/topics/r80401seika_shimizu
- https://pubs.acs.org/doi/10.1021/jacs.5c22771
七員環を埋め込んだ縮環ルブレン誘導体:その曲率に依存した光物理特性および半導体特性 Fused Rubrene Derivatives with Embedded Seven-Membered Rings: Curvature-Dependent Photophysical and Semiconductor Properties
Masato Hisada,Kirill Bulgarevich,Yusuke Tsutsui,Kiyoshi Miyata,Daiki Shimizu,Shu Seki,Kazuo Takimiya,Kenji Matsuda
Journal of the American Chemical Society Published: February 8, 2026
DOI:https://doi.org/10.1021/jacs.5c22771
Abstract
The synthesis and characterization of a fused rubrene derivative with two embedded seven-membered rings are described. Owing to the incorporation of heptagons, fused rubrene was obtained as two isolable conformers in the C2h-symmetric saddle and D2-symmetric twisted structures. The isomers were both highly curved, as judged from the mean plane deviation of 1.10 Å (saddle) and 1.15 Å (twist). Kinetic studies using NMR spectroscopy and DFT calculations revealed that the saddle isomer was 11.0 kJ·mol–1 more stable at 25 °C than the twisted isomer. The isomers were kinetically stable under ambient conditions but interconverted at high temperatures with an isomerization barrier of 136.2 kJ·mol–1 at 25 °C from the twist to the saddle isomer. Both isomers displayed narrower optical band gaps than the nonfused rubrene. The two isomers exhibited optical properties that depended on their different molecular curvatures, which are thoroughly addressed in this work. The fused structure also enhanced the photostability compared with that of rubrene. Transient absorption studies suggested fast (τ = 7.2 ps) singlet fission of the twisted isomer in a thin film upon photoirradiation. Single-crystal OFET measurements demonstrated that the hole mobility of the crystalline saddle isomer is comparable to that of representative PAHs employed as organic semiconductors.



