2023-09-07 アルゴンヌ国立研究所(ANL)
◆リチウム硫黄電池はリチウムイオン電池に比べてエネルギー密度が高く、コストが低く、重要な資源を必要としないため、優れた特性を持つが、商業用途での性能低下が課題であった。
◆科学者たちは触媒を用いて硫黄の損失問題を解決し、その反応メカニズムを解明した。これにより商業用途での性能が向上し、持続可能な輸送システムへの一歩が踏み出せる可能性がある。この研究は、次世代電池にも適用可能かどうかも調査され、環境に優しい未来に向けた重要な進展である。
<関連情報>
- https://www.anl.gov/article/previously-unknown-pathway-to-batteries-with-high-energy-low-cost-and-long-life
- https://www.nature.com/articles/s41586-023-06326-8
- https://www.nature.com/articles/s41565-020-00797-w
リチウム硫黄電池の界面集団反応挙動を可視化 Visualizing interfacial collective reaction behaviour of Li–S batteries
Shiyuan Zhou,Jie Shi,Sangui Liu,Gen Li,Fei Pei,Youhu Chen,Junxian Deng,Qizheng Zheng,Jiayi Li,Chen Zhao,Inhui Hwang,Cheng-Jun Sun,Yuzi Liu,Yu Deng,Ling Huang,Yu Qiao,Gui-Liang Xu,Jian-Feng Chen,Khalil Amine,Shi-Gang Sun & Hong-Gang Liao
Nature Published06 September 2023
DOI:https://doi.org/10.1038/s41586-023-06326-8
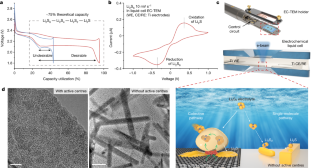
Abstract
Benefiting from high energy density (2,600 Wh kg−1) and low cost, lithium–sulfur (Li–S) batteries are considered promising candidates for advanced energy-storage systems1,2,3,4. Despite tremendous efforts in suppressing the long-standing shuttle effect of lithium polysulfides5,6,7, understanding of the interfacial reactions of lithium polysulfides at the nanoscale remains elusive. This is mainly because of the limitations of in situ characterization tools in tracing the liquid–solid conversion of unstable lithium polysulfides at high temporal–spatial resolution8,9,10. There is an urgent need to understand the coupled phenomena inside Li–S batteries, specifically, the dynamic distribution, aggregation, deposition and dissolution of lithium polysulfides. Here, by using in situ liquid-cell electrochemical transmission electron microscopy, we directly visualized the transformation of lithium polysulfides over electrode surfaces at the atomic scale. Notably, an unexpected gathering-induced collective charge transfer of lithium polysulfides was captured on the nanocluster active-centre-immobilized surface. It further induced an instantaneous deposition of nonequilibrium Li2S nanocrystals from the dense liquid phase of lithium polysulfides. Without mediation of active centres, the reactions followed a classical single-molecule pathway, lithium polysulfides transforming into Li2S2 and Li2S step by step. Molecular dynamics simulations indicated that the long-range electrostatic interaction between active centres and lithium polysulfides promoted the formation of a dense phase consisting of Li+ and Sn2− (2 < n ≤ 6), and the collective charge transfer in the dense phase was further verified by ab initio molecular dynamics simulations. The collective interfacial reaction pathway unveils a new transformation mechanism and deepens the fundamental understanding of Li–S batteries.
両末端結合部位を有するマクロポーラス触媒正極を介した高エネルギー・長サイクルのリチウム硫黄パウチ電池 A high-energy and long-cycling lithium–sulfur pouch cell via a macroporous catalytic cathode with double-end binding sites
Chen Zhao,Zhou Yu,Leicheng Zhang,Inhui Hwang,Yu-Xue Mo,Yuxun Ren,Lei Cheng,Cheng-Jun Sun,Yang Ren,Xiaobing Zuo,Jun-Tao Li,Shi-Gang Sun,Khalil Amine & Tianshou Zhao
Nature Nanotechnology Published:23 November 2020
DOI:https://doi.org/10.1038/s41565-020-00797-w
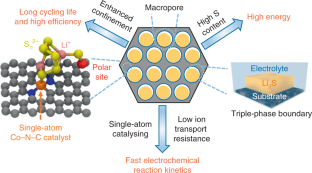
Abstract
Lithium–sulfur batteries are attractive alternatives to lithium-ion batteries because of their high theoretical specific energy and natural abundance of sulfur. However, the practical specific energy and cycle life of Li–S pouch cells are significantly limited by the use of thin sulfur electrodes, flooded electrolytes and Li metal degradation. Here we propose a cathode design concept to achieve good Li–S pouch cell performances. The cathode is composed of uniformly embedded ZnS nanoparticles and Co–N–C single-atom catalyst to form double-end binding sites inside a highly oriented macroporous host, which can effectively immobilize and catalytically convert polysulfide intermediates during cycling, thus eliminating the shuttle effect and lithium metal corrosion. The ordered macropores enhance ionic transport under high sulfur loading by forming sufficient triple-phase boundaries between catalyst, conductive support and electrolyte. This design prevents the formation of inactive sulfur (dead sulfur). Our cathode structure shows improved performances in a pouch cell configuration under high sulfur loading and lean electrolyte operation. A 1-A-h-level pouch cell with only 100% lithium excess can deliver a cell specific energy of >300 W h kg−1 with a Coulombic efficiency >95% for 80 cycles.


