2026-03-04 ウィスコンシン大学マディソン校 (UW-Madison)
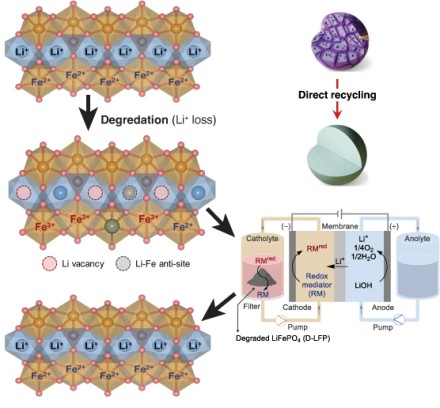
Schematic of LFP degredation and regeneration process. Loss of lithium ions creates vacancies in the crystal structure, limiting its ability to hold a charge. Direct recycling replaces the lost ions in the structure.
<関連情報>
- https://energy.wisc.edu/news/new-life-spent-batteries-chemistry-has-solution
- https://onlinelibrary.wiley.com/doi/10.1002/anie.202520213
使用済みLiFePO4電池正極の酸化還元電気化学的再生 Redox-Mediated Electrochemical Regeneration of Spent LiFePO4 Battery Cathodes
Deok-Ho Roh, Dayun Jung, James B. Gerken, Jesse J. Martinez, Eric Kazyak, Shannon S. Stahl
Angewandte Chemie International Edition Published: 10 February 2026
DOI:https://doi.org/10.1002/anie.202520213
ABSTRACT
Direct recycling of lithium-ion battery cathodes offers considerable appeal over metallurgical approaches. Here, we demonstrate a mediated electrochemical method for direct regeneration of degraded LiFePO4 (LFP). The approach uses a redox mediator, iron propylenediamine tetraacetate, that undergoes electrochemical reduction and is circulated through an external reservoir, where it supplies the electrons needed to regenerate LFP in the presence of Li+ ions derived from LiOH oxidation. Rapid outer-sphere electron transfer is observed from the mediator to the degraded LFP material. This feature, together with good aqueous solubility of the mediator (0.3 M), supports current densities up to 100 mA/cm2, and this electrochemical recycling process is demonstrated on 100 g scale. 57Fe Mössbauer spectroscopy is used to monitor the correction of structural defects in the degraded LFP, providing the basis for regeneration of LFP that matches the electrochemical performance of pristine LFP.